
In the EAGLE-1 Phase III trial, gepotidacin met the primary efficacy endpoint of non-inferiorty to the current leading treatment for uncomplicated urogenital gonorrhea.

In the EAGLE-1 Phase III trial, gepotidacin met the primary efficacy endpoint of non-inferiorty to the current leading treatment for uncomplicated urogenital gonorrhea.
BAY 2927088 is an oral, reversible small molecule tyrosine kinase inhibitor being analyzed for the treatment of unresectable or metastatic non-small cell lung cancer with tumors that have activating HER2 mutations.
Survodutide, a GLP-1 receptor dual agonist with a novel mechanism of action, was the first treatment to produce findings this significant in a Phase II trial of metabolic dysfunction-associated steatohepatitis.
The FDA has assigned a supplemental Biologics License Application submitted by Regeneron and Sanofi for Dupixent in the treatment of COPD with type 2 inflammation with a PDUFA date of June 27, 2024.

SKYTyphoid showed a positive immunogenicity and safety profile compared to other polysaccharide-protein conjugate typhoid vaccines that obtained prequalification certification by the World Health Organization.

Early-stage trial results indicate that NLRP3 inflammasome inhibitors were able to achieve nearly the same weight loss as Wegovy while also reducing inflammatory biomarkers linked to heart disease.
Ocifisertib is a first-in-class, novel PLK4 inhibitor that has demonstrated significant activity as a monotherapy in both solid and liquid tumors.

Kaufman discusses the ways the digital biomarkers are improving Alzheimer’s research by directly tackling some of the unique challenges that researchers face.

While no company has yet achieved the full promise of AI to radically accelerate time to market, meaningful strides are being made across the drug discovery funnel.

The FDA assigned the biologics license application for linvoseltamab to treat relapsed/refractory multiple myeloma with a Prescription Drug User Fee Act of August 22, 2024.
Injectable long-acting Cabenuva (cabotegravir + rilpivirine) produced superior efficacy in viral load suppression compared with daily oral antiretroviral therapy in patients with HIV who have adherence challenges.

TEV-’574 has shown potential as a treatment for inflammatory bowel diseases such as ulcerative colitis and Crohn disease.
The FDA assigned the supplemental new drug application for Krazati (adagrasib) plus cetuximab in patients with locally advanced or metastatic colorectal cancer with a Prescription Drug User Fee Act goal date of June 21, 2024.
The FDA set a Prescription Drug User Fee Act date during Q4 of 2024 for the biologics license application for datopotamab deruxtecan in patients with previously treated advanced nonsquamous non-small cell lung cancer.
Positive Phase III study results for Tonmya (cyclobenzaprine HCl sublingual tablets) will be basis of New Drug Application to the FDA for the management of fibromyalgia.

Looking to hold an advisory board? Here are a few suggestions.
As therapeutics become more personalized and targeted, technology, such as open-source software, can help companies innovate.
BXCL701 is an oral innate immune activator being investigated in combination with Keytruda (pembrolizumab) in patients with metastatic small cell neuroendocrine prostate cancer.
In clinical trials, bepirovirsen showed the potential to address a significant unmet medical need for patients with chronic hepatitis B by reducing hepatitis B surface antigen levels and HBV DNA.

Novel oral orexin receptor 2 agonist produced statistically significant and clinically meaningful improvements in wakefulness compared with placebo in patients with narcolepsy type 1.

Researchers evaluate a potential association between the use of phosphodiesterase type 5 inhibitors for erectile dysfunction and a reduced risk of Alzheimer disease.
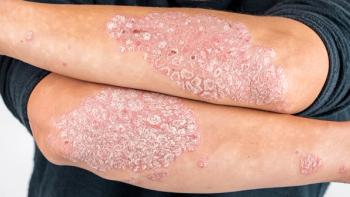
JNJ-2113, an oral IL-23–receptor antagonist peptide, showed consistency across clinician and patient-reported outcomes in patients with moderate-to-severe plaque psoriasis.

Data from the ENHANCE-3 trial of magrolimab in combination with azacitidine plus Venclexta showed futility and an increased risk of death in patients with acute myeloid leukemia.
The biopharma company’s Phase 3 trial for its acoramidis product generated statistically significant results.

The Maryland facility will create cell therapies for use in future cancer trials.